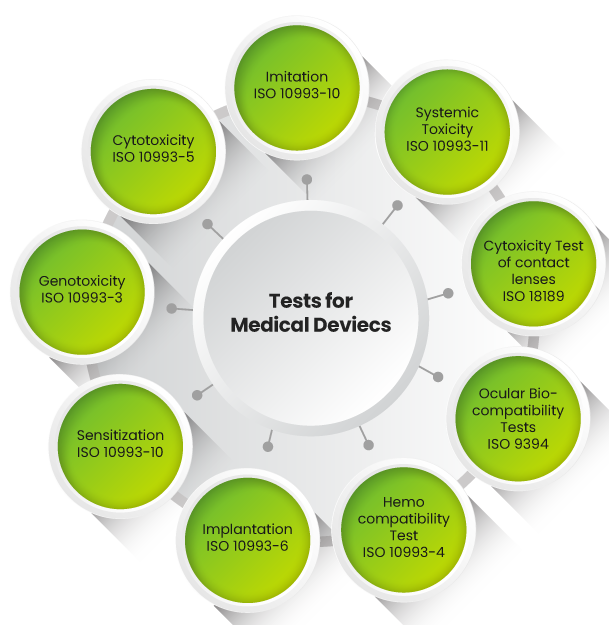
Biocompatibility Testing of Medical Devices is required to establish the safety of medical devices and is a critical part of the regulatory approval process. Leveraging on its Toxicology and Safety experience and credibility of over three decades, Adgyl Lifesciences is your ideal partner to provide Biocompatibility testing services toward the development of your Medical Device programs, The data can be used for 510K submissions, and to various regulatory and notified bodies. The Test Facility, Adgyl Lifesciences, is GLP certified since 1992, AAALAC certified since 2001, and is an ISO 17025 certified and Central Drugs Standard Control Organization (CDSCO) registered facility.
The Adgyl Lifesciences’ Medical Device Testing team has the expertise and experience of 30 years of GLP Testing, to deliver superior quality work at a competitive price and in a time-effective manner. The Team, with an accumulated experience of 100+ years in Biological safety assessment, Biocompatibility testing strategy and evaluation of toxicological results. can help develop -an appropriate testing program for assessing the safety of – Medical Devices as per the latest ISO 10993 standards, FDA guidance and other guidelines, and are performed in compliance with GLP to ensure worldwide acceptability.
*: Implantation of large animals is performed at a partner facility